Winter 2020 edition: Sports Medicine for the Primary Care Provider

The Winter 2020 edition of Sports Medicine for the Primary Care provider was recently published. Articles in this edition include “Platelet-Rich Plasma Injections,” “Sleep and the Athlete,” “Musculoskeletal Ultrasound and Athletes” and “Research in Brief.”
A printable version of this newsletter is available here.
All articles also appear below.
Welcome
Dear Fellow Health Care Provider,
Welcome to the latest edition of our primary care sports medicine newsletter, a biannual publication focusing on seasonal sports topics. We hope you find the information useful and appreciate any feedback you have to enhance our efforts.
For questions, feedback and inquiries about future issues, please contact Tracie Kirkessner at tkirkessner@pennstatehealth.psu.edu or me at msilvis@pennstatehealth.psu.edu.
Enjoy,
Matthew Silvis, MD
Professor, Departments of Family and Community Medicine and Orthopaedics and Rehabilitation
Penn State Health Milton S. Hershey Medical Center
Platelet-Rich Plasma Injections
By Shawn Phillips, MD
Platelet-rich plasma (PRP) injections, while not new, have become a more popular treatment for many conditions in orthopedics and sports medicine. Due to the increased popularity, patients have become more interested in learning more about the potential benefits PRP injections might offer them.
The following information, including indications and uses of PRP in sports medicine, is created to help guide such conversations with patients.
PRP injections are used by physicians to treat pain in joints or tendons that have not responded to more conservative treatment, such as physical therapy, cortisone injections, antiinflammatory medications and bracing.
PRP is obtained from a patient’s own blood. Once the blood is drawn, it is placed in a centrifuge to isolate the PRP component. After spinning is complete and the component is isolated, it has a higher concentration of platelets and other growth factors. Recent advances in technology allow physicians to adjust the centrifuge to alter the level of PRP concentration to a higher or lower number of leukocytes. Research has demonstrated that PRP with different concentrations of leukocytes may have benefits for different orthopedic conditions.
Treatment for Tendon Pain
 For several decades, PRP has been an option to treat chronic tendon pain that has not responded to other therapies. More recently, musculoskeletal ultrasound has allowed physicians to evaluate and more accurately inject injured or degenerative tendons, increasing the utilization of PRP in chronic tendon pain.
For several decades, PRP has been an option to treat chronic tendon pain that has not responded to other therapies. More recently, musculoskeletal ultrasound has allowed physicians to evaluate and more accurately inject injured or degenerative tendons, increasing the utilization of PRP in chronic tendon pain.
The goal of PRP when injected into an injured tendon is often to irritate the tendon to restart the inflammatory cascade. This is thought to promote the potential for healing within the tendon. Some research studies have indicated that leukocyte-rich PRP is more effective than other forms of PRP when injected into an injured or degenerative tendon, likely because of the increased potential for inflammation.
After PRP is injected, patients often resume physical therapy designed to load the tendon. Patients should be warned that pain usually increases for several days immediately after the injection. To ensure the inflammatory process continues, NSAIDS should be avoided for pain control, at this stage. However, acetaminophen and ice can be used for pain control. A sling or brace may also be worn for comfort initially after injection.
Treatment for Arthritis Pain
More recently, intra-articular injections with PRP have been performed to help patients with pain from osteoarthritis or other intra-articular conditions. As inflammation is usually not desired after an intra-articular injection, leukocyte-poor PRP is often chosen for these cases. Preliminary research has shown that PRP is equal to or better than corticosteroid and Visco supplementation injections to control pain secondary to arthritis.
At one time, there was hope that PRP may help replenish or regenerate injured cartilage. Unfortunately, this has not been shown to be true, but there does appear to be a role for PRP in pain control in arthritis. Unlike tendon injections, intra-articular PRP injections do not usually increase pain initially, and patients can resume usual activities the day after their injection. There is not sufficient data to say whether PRP injections should be performed as a single injection or in a series.
While not brand-new, PRP is still an up-and-coming modality in sports medicine and orthopedics. For this reason, many insurance companies do not cover the cost of a PRP injection, and there will likely be an out-of-pocket cost for patients who wish to consider PRP.
While much more research is needed, PRP may be a reasonable option to consider for some patients with chronic joint or tendon pain that has not responded to other treatments.
Sleep and the Athlete
By Joseph Andrie, MD, Primary Care Sports Medicine fellow
It is already well-established that a good night’s sleep is essential for overall health and well-being at any age. In addition, adequate sleep for athletes is of utmost importance. What is considered “adequate” sleep for athletes at any level versus non-athletes differs.
As a general population, athletes overall do not achieve healthy sleep patterns. In a study that examined more than 14,000 National Collegiate Athletic Association (NCAA) athletes, fewer than 25% reported eight hours or more of sleep on a typical night. Furthermore, 61% of this group reported daytime fatigue greater than three times per week.
Poor sleep is associated with various challenges that could be particularly difficult for athletes:
- Delayed reaction time
- The perception of increased effort necessary to complete tasks
- Feeling exhausted more quickly
- Increased mood disturbances
In healthy adolescents, sleeping fewer than seven hours per night for only a week leads to greater anxiety and decreased emotional regulation. Adolescents who reported fewer than eight hours of sleep per night were 1.7 times more likely to suffer from a musculoskeletal injury and more likely to present with a medical illness when training load and intensity increased.
The two most common sleep-related diagnoses in athletes are circadian dysregulation and insomnia.
Circadian dysregulation is most common in athletes who frequently travel across time zones. This diagnosis can negatively impact metabolism, performance and psychological functioning. Practices and training sessions in the early morning can also lead to impaired sleep and decreased performance outcomes as each athlete has a unique chronotype for their personal peak performance time.
Medical interventions to aid with circadian dysregulation include the use of melatonin, as well as timed bright light exposure therapy.
For athletes with true insomnia beyond a circadian dysregulation, recommendations include cognitive-behavioral therapy, as well as medications from the imidazopyridine class, such as zolpidem and zopiclone.
For any individual with poor sleep patterns, sleep apnea should be considered as an underlying diagnosis, which is well known to impair athletic performance, if untreated.
To help primary care physicians address these issues with their athlete patients, the British Journal of Sports Medicine and the NCAA both published recommendations and guidelines pertinent to sleep disorders in athletes. These recommendations are highlighted below. In addition, it helps to recommend common remedies, as one would share with the general population.
Sleep screening should be a priority for athletes. At a time when early sports specialization and elite-level play at younger ages has become an issue, sleep deprivation and disturbed sleep need to be addressed to avoid overtraining and to maximize training gains through adequate sleep. As primary care and sports medicine physicians, we may want to consider the addition of sleep screening to our preparticipation exams. Furthermore, we need be prepared to address any sleep concerns in the context of the athlete, whether at the youth, collegiate or professional levels.
Potential Solutions to Disordered Sleep in the Athlete
- Tracking and monitoring sleep
- Modeling healthy sleep by coaches
- Encouraging healthy sleep as part of the training protocol
- Promoting sleep health education
- Scheduling training around sleep and circadian rhythms
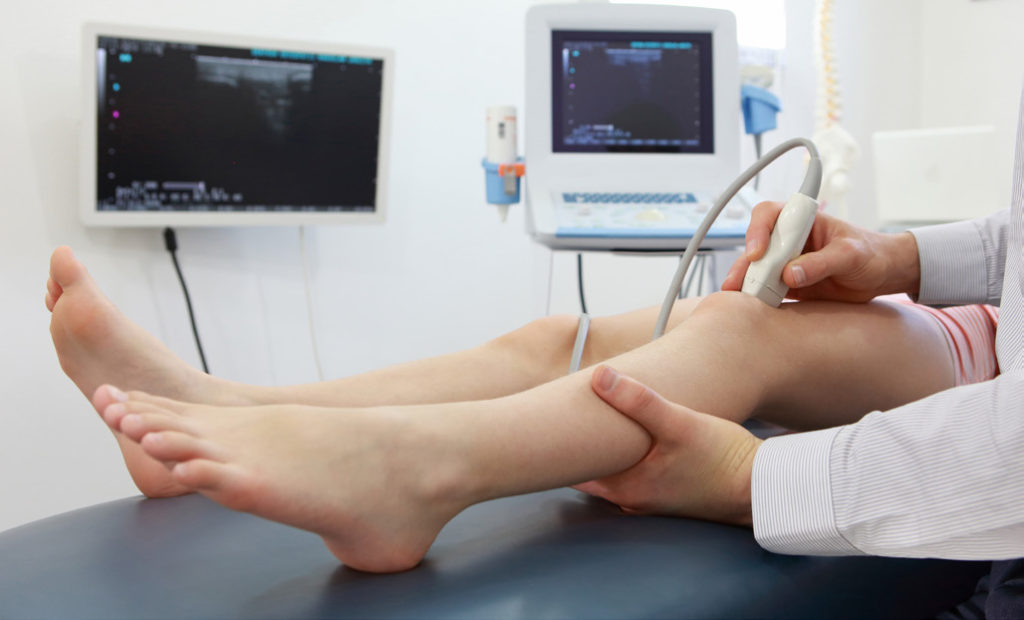
Musculoskeletal Ultrasound and Athletes
By Philip Bosha, MD, and Peter Seidenberg, MD, State College faculty
Participation in sports carries numerous benefits but also includes the risk of injuries. Sports medicine physicians often use musculoskeletal ultrasound in the evaluation or treatment of injuries as an efficient and effective tool. This low-cost modality can be performed in the office or even the athletic training room and does not expose athletes to radiation.
Sports medicine physicians often utilize ultrasound as an extension of physical examinations, when appropriate, to help determine the cause of pain. Shoulder pain, for instance, is common in overhead athletes, such as swimmers and throwers. Often the initial examination may be inconclusive, but ultrasound may aid in making a more specific diagnosis, including whether the rotator cuff tendons or bursa are involved. This can help guide treatment, including specific recommendations for physical therapy referral.
Ultrasound can also be used to perform a guided corticosteroid injection to help with pain.
Many young athletes are involved in baseball and softball in the spring and fall seasons. Medial elbow pain is a common complaint among throwing athletes. Concerned parents may be worried that this will lead to Tommy John surgery. Ultrasound is a very useful diagnostic tool to evaluate medial elbow pain. Unlike other types of imaging, dynamic testing can be performed by placing valgus stress on the elbow similar to what happens in throwing, and the ulnar collateral ligament can be assessed. More commonly, the wrist flexor muscles cause medial elbow pain and can be evaluated more accurately with ultrasound.
This winter, you may have athletes playing ice hockey. Hip and groin pain are common complaints among skaters. Musculoskeletal ultrasound is effective to evaluate and diagnose many sources of hip pain, including adductor muscle strains, femoro-acetabular impingement, snapping hip syndrome and iliopsoas bursitis. Ultrasound can be used to perform hip joint injections to both help with pain and aid in diagnosis. This avoids the need to refer the patient to interventional radiology for a fluoroscopic-guided injection and avoids additional radiation exposure. The assessment of hip pain and the ultrasound-guided injection can typically be handled at the same appointment, an additional convenience for the patient.
Ultrasound may also be used to assess the amount of damage in thigh injuries. Direct trauma to the thigh may cause an accumulation of blood within the muscle that calcifies over time. Common injuries include taking a football helmet to the side of the upper leg or getting hit by a lacrosse ball. Ultrasound can be used to determine the extent of muscle damage, how much blood has accumulated in the area and when the injury has healed.
Ultrasound can also quickly assess the exact location of a hamstring strain. If the injury is in the muscle, conservative management is indicated. On the other hand, if the tendon has avulsed from the ischial tubescosity, surgery may be necessary.
Ultrasound can have several uses in the evaluation of a patient with a sports injury. It is a tool to provide patients with an efficient diagnosis and treatment, allowing return to the field of play as quickly and safely as possible.
Research in Brief
Saliva microRNA for Concussion Assessment
2020 AMSSM Research Submission
 Authors: Onks C, Loeffert J, Loeffert A, Olympia R, Kim R, Zhang K, Hicks S
Authors: Onks C, Loeffert J, Loeffert A, Olympia R, Kim R, Zhang K, Hicks S
Affiliation: Penn State College of Medicine (Departments of Pediatrics, Family Medicine, Emergency Medicine), Hershey, Pa.
Purpose: There is no objective, biologic tool to detect concussion or guide clinical management. The purpose of this study was to validate the utility of saliva miRNA and define longitudinal trends during the recovery period.
Methods and study design: We collected saliva and symptom burden (SCAT-5) from 150 individuals (7-21 years) with a clinical concussion diagnosis across 0-2, 3-6, 7-14, 15-29, and 30-60 days post-injury. Saliva miRNA levels within the 443 concussion samples were measured with RNA sequencing and compared to 218 samples from age and sex-matched controls (including healthy and post-exercise participants).
Results: Fifteen miRNAs differed in concussion patients compared to control on Kruskal-Wallis testing (FDR < 0.05). Within the concussion participants, all 15 miRNAs trended back toward control levels by 30-60 days post injury. Logistic regression employing six of the 15 miRNAs differentiated control and concussion participants with an area under the curve (AUC) of 0.78 in a training set (n = 244) and 0.84 in a naïve test set (n=24). Similarly, six miRNAs were able to differentiate symptomatic (SCAT-5 symptom score > 7) and asymptomatic concussion participants with an AUC of 0.73 in a training set (n = 219) and 0.76 in a naïve test set (n=44). Furthermore, five miRNAs showed significant (R > 0.3; FDR < 0.05) associations with subjective or objective measures of concussion-related symptoms including headache, feeling slowed down, reaction time, dizziness and balance. Conclusions: Saliva miRNAs levels are altered in children with concussion and display predictable longitudinal trends following injury. Saliva miRNA levels display promise as an objective aid to diagnosis, a molecular signature for brain recovery and a biologic measure of concussion phenotype.
Significance: Saliva miRNA measurement represents a noninvasive, objective tool that could be rapidly assessed to provide biologic evidence for clinicians managing pediatric concussion.
Acknowledgements: Funding for this study was provided by a research agreement between Penn State and Quadrant Biosciences Inc. Dr. Hicks serves as a paid consultant for Quadrant Biosciences. We thank Jennifer Stokes and Molly Carney for assistance with sample collection.
Contact Us
Learn more about the Sports Medicine team at Penn State Health here.
If you're having trouble accessing this content, or would like it in another format, please email Penn State Health Marketing & Communications.
